Over the last 100 years, the advancement of medical science has affected all of our lives in immeasurable ways. It has allowed researchers to eliminate killer viruses like Smallpox and cripplers like Polio. And, thanks to the wonders of modern science, doctors were able to reduce AIDS from a virtual death sentence to a manageable chronic disease. All these medical miracles have been made possible through intense medical research studies. These studies need to do a few things very well, including count and measure. A lot goes into ensuring the accuracy and validity of the scientific tests and the results of those studies. Time is usually of the essence, and any slight deviations could lead the experts down the wrong path, possibly delaying the treatments people need immediately.
See how we helped a group of researchers, investigators, and advocates who evaluate the impact of AIDS on mothers and their children worldwide improve the workflows of their clinical studies. These dedicated professionals represent a few different organizations and institutions, but they share the common goal of treating and preventing the transmission of the human immunodeficiency virus (HIV), the infection that causes AIDS. And a lot of their work is based on meticulous clinical trials.
Planning Work
Running a medical study can be a complicated and time-consuming project. A good deal of preparation work is required just to get approval for a clinical trial, and then there's more work to be done during the trial. Investigators usually spend long hours filling out paperwork, inspecting sites, labs, and equipment, and organizing virtual piles of discrete spreadsheets before a clinical trial even starts.
The team we worked with understands the challenges of research studies all too well. They have to coordinate the logistics of medical research studies conducted in dozens of clinical sites and laboratories across the globe. The investigators rely on several different factors to ensure a successful study, including their equipment, which has to be in perfect working order.
The researchers face the daunting task of confirming that each of the labs within all their clinical sites has the right equipment to process and test their samples. Then they need approval from a third-party board, Institutional Review Boards, to run the required tests. This process involves gathering information from each of the labs, and of course, each lab has its own way of sharing those details depending on the preferences of the person providing the data. They receive reports in a variety of formats, from Excel and CSV to PDFs. PDFs are difficult to edit and therefore require manual extraction, which means additional work for the investigators. It would be helpful if the data were collected in a standard format as it would certainly speed up the study's launch.
Procedural Work
With that understanding, The Support Group sat down with the investigators and the labs to strategize a plan for creating workflows that all the parties could use to ensure their data was formatted uniformly and entered correctly. The Support Group utilized a workplace innovation platform, Claris FileMaker, specifically the FileMaker WebDirect technology, to allow the labs to input the data directly into a centralized system via a web browser. This cloud-based system allows the labs to directly enter their data into a streamlined repository and apply specific business logic to validate it in real-time. So, the person at the lab entering the information receives immediate feedback, thereby making it possible to make adjustments right on the spot. Real-time responsiveness saves lots of precious time. In addition, accessibility is vital to global clinical research studies. The web and the cloud seamlessly make this system available worldwide, from the United States to South Africa, Brazil, and Thailand.
Data accuracy was the primary objective of this custom solution. But usability was certainly top of mind as well. The workflows and interface had to support the users without hindering them or the process in any way. Otherwise, the system would be counterproductive. We thoroughly considered how the workflows affected user productivity by testing extensively in the field. Our FileMaker developers quickly integrated user feedback to improve the interface and overall experience with the tool.
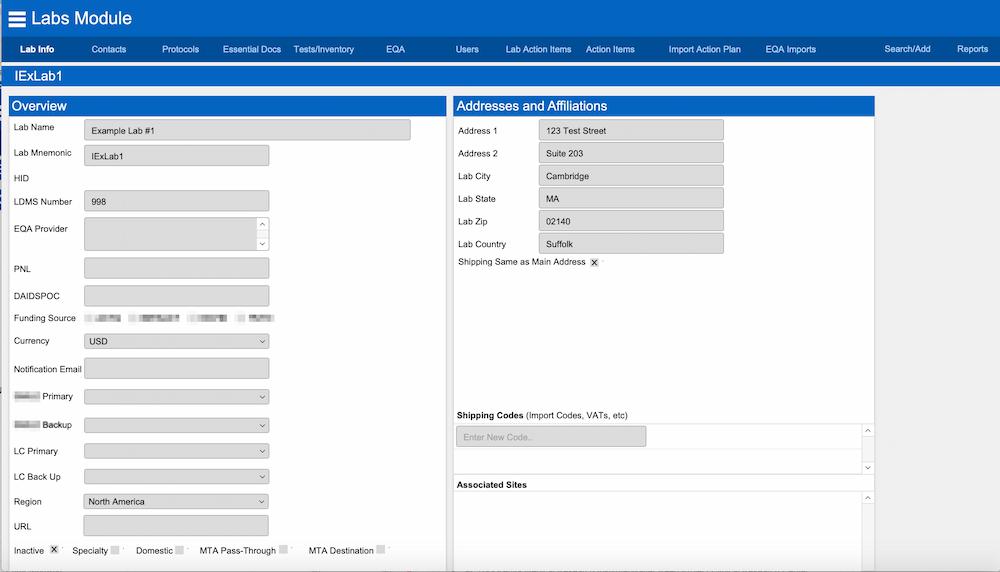
FileMaker medical research custom application
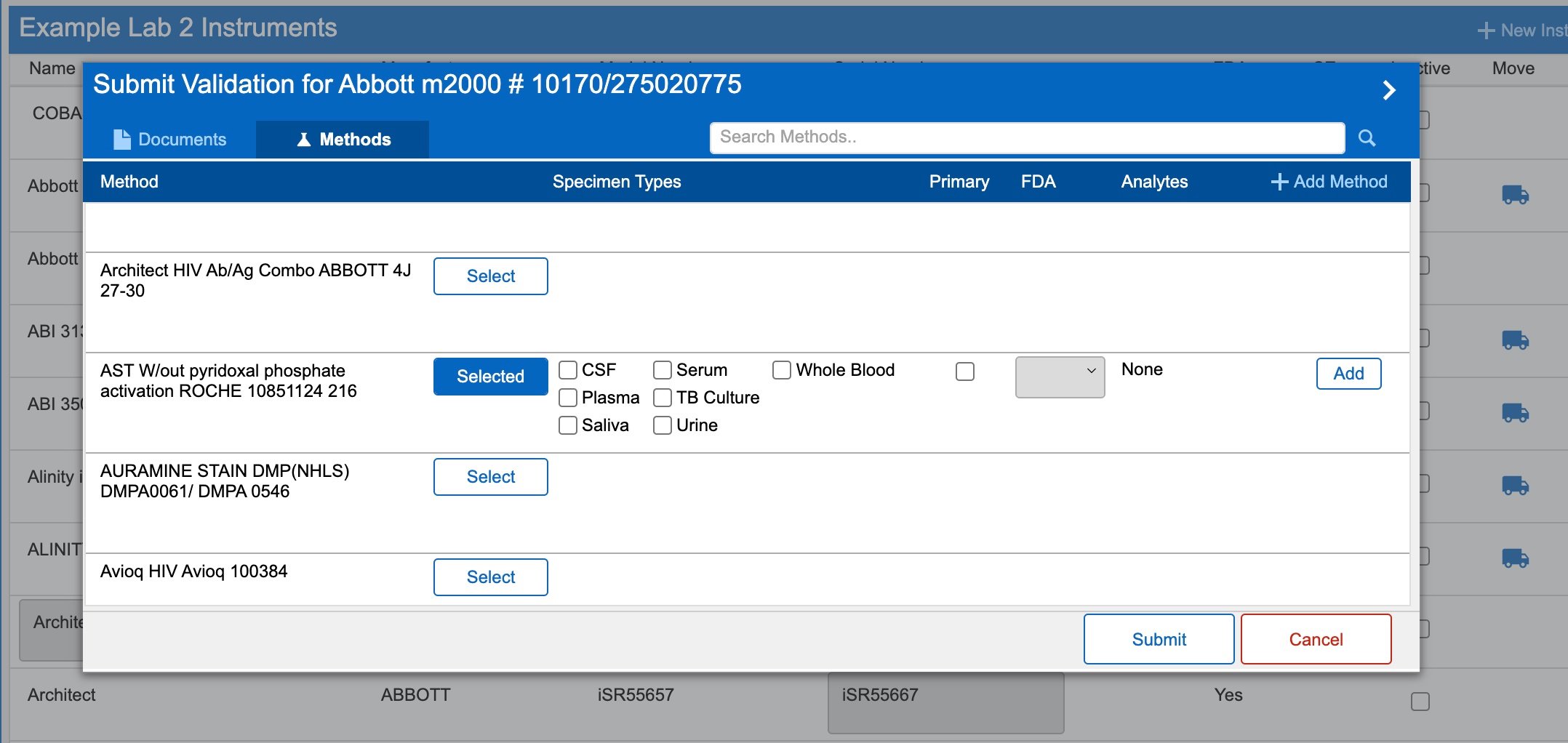
FileMaker medical research custom application user interface
Generally, there are lower expectations in terms of performance for web-based applications versus desktop applications. However, that was not the case for this application. This particular FileMaker WebDirect technology that runs in a standard web browser gave the users in the remote labs nearly the same user experience and speed as the users in the home office running the FileMaker desktop client. Another time-saving benefit.
Problem-Solving Work
These workflow improvements and many others related to data analysis and cost determination significantly streamlined the research preparation process. What used to take several months to do now only takes about a week or so. Although it's always nice to reduce administrative work and automate number crunching, the real reward is that the application allows more clinics to get online faster than before so that these essential studies can ramp up quickly. Ultimately it means samples and specimens are efficiently provided to the scientists on the frontlines of the fight to keep mothers and their babies who are struggling with HIV infection healthy and alive.
Working with the team at The Support Group (TSG) has far exceeded our expectations! The work our organization does is both specialized and full of technical content, so we weren’t sure how well this would translate when describing our vision for the database to a FM Developer. TSG took the time to meet and plan out the design of the database and create a framework that has allowed us to significantly expand our operations and increase our productivity. In our multi-year partnership with TSG, each developer has managed to create a successful solution for every design solution that we have thrown at them!
We've been developing custom apps for more than 30 years. See how we helped one of our clients develop a reporting application and another client track inventory efficiently. Read more of our Customer Success Stories and case studies. Feel free to contact us to learn more about our custom software development services or schedule a free initial consultation.



